Research
Science
Extracellular Matrix (“ECM”) is the non-cellular component within all tissues and organs which not only provides essential physical structure for the cellular constituents, but also initiates essential biochemical and biomechanical signals that are required for tissue development, changes, and stability.
In the gastrointestinal tract, the layer immediately below the epithelium is known as the basement membrane. This basement membrane is composed of extracellular matrix.
ECM is isolated from tissues and organs by processes that remove the cells in a process called decellularization.
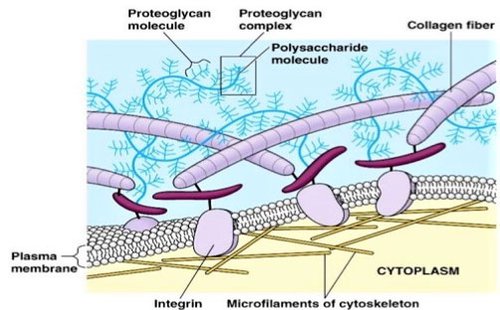
The ECM contains 3 classes of molecules:
- Structural proteins (collagens and elastins)
- Protein-polysaccharide complexes to embed the structural proteins (proteoglycans)
- Adhesive glycoproteins to attach cells to matrix (fibronectins and laminins)
In the colon, the extracellular matrix provides a barrier and structural support for the epithelial lining of the gastrointestinal tract.
Although, fundamentally, ECM is composed of water, proteins and polysaccharides, and each tissue has an ECM with a unique composition and topology (spatial arrangement and orientation). This uniqueness is generated during tissue development through a dynamic and reciprocal biochemical and biophysical communication between the various cellular components (e.g. epithelial, fibroblast, adipocyte, endothelial elements), and the evolving cellular and protein microenvironment.
The ECM functions as a physical barrier, an anchorage site, or a movement track for cell migration. The physical properties of the ECM, including its rigidity, density, porosity, insolubility (resistance to being dissolved in liquid) and topography, provide physical cues to the cells.
 Technology
Technology
The Regentys™ Extracellular Matrix Hydrogel (“ECMH”) is a proprietary, porcine-derived, small intestine submucosa (SIS) extracellular matrix product that is rectally administered. The ECMH is a powder that is reconstituted with saline and delivered as a liquid.
As ECMH reaches body temperature, it gels and coats the mucosal lining of the GI tract. This provides a physical barrier and a bio-scaffold to help the body rebuild the damaged mucosal lining.
Research
Regentys™ has completed multiple preclinical studies. The results of the pivotal preclinical study have been presented at multiple major scientific and clinical meetings, and are published in the University of Oxford’s Journal of Crohn’s and Colitis (available here). The paper, published by Dr. Timothy Keane, PhD found that ECMH:
- Restores cell barrier function
- Promotes rapid tissue regeneration
- Reduces clinical symptoms
- Reverses inflammation and ulceration
- Changes biomarkers
In simple terms, all of these functions allow ECMH to help the colon lining heal and possibly maintain its resistance to future active disease.
Regentys™ plans to begin human trials in the middle of 2018, initiating a pilot study in patients with mild to moderate ulcerative colitis. Future applications of the Regentys technology may be applied to other GI diseases such as more extensive UC, rectal mucositis, or Crohn’s Disease.